Absorption, Metabolism, Excretion, and Health Outcomes of Sugar Alcohols.
Absorption, metabolism, excretion, and health outcomes of various sugar alcohols.
Department of Standards, Paleo Foundation, Encinitas, CA
Abstract
Food manufacturers routinely use sugar alcohols as substitutes for sugar due to their low calorie content and other health benefits. However, differences in absorption, metabolism, and excretion of various sugar alcohols can alter the net carbohydrate calculations of low-carbohydrate foods, especially in the context of a ketogenic diet and Keto Certified products. This review presents evidence that sugar alcohols should be calculated based on their respective absorption, metabolism, and excretion rates, and discusses the safety and health benefits of sugar alcohols in the context of sugar replacements.
KEYWORDS
Sugar Alcohols, Polyols, Erythritol, Xylitol, Mannitol, Sorbitol, Maltitol, glycerol, glycerin.
Food manufacturers routinely use sugar alcohols as substitutes for sugar due to their low-calorie content and other health benefits. While there is no doubt that sugar alcohols can provide advantages over plain sugar [1], there are still many misconceptions that remain about these compounds, which at best, lead to harmless myths and at worst, are incredibly dangerous. Sugar alcohols, also known as ‘polyols’, are a subgroup of carbohydrates characterized by the presence of an -CH-OH (alcohol) group in the area where a C=O (carbonyl) group is typically found [1]. Thus, from a biochemical perspective sugar alcohols are not actually sugars despite the misleading name. The distinction between the two is made clearer by the legal definition of sugars, monosaccharides or disaccharides [2]. However, polyols encompass a wider range of carbohydrates and can be mono-, di-, oligo-, and polysaccharides. Most sugar alcohols can be recognized by the presence of the “-itol” at the end of the name and some of the most commonly used polyols in food products are erythritol, xylitol, sorbitol, maltitol, mannitol, and glycerol. Although sugar alcohols can be found naturally in some plants and plant foods such as birch trees and berries, the vast majority are now produced via hydrogenation (the addition of hydrogen in a chemical reaction) of sugars with certain catalysts to speed up the reaction or they are produced via fermentation of sugars [1,3].
2 | PROPERTIES OF SUGAR ALCOHOLS
ABSORPTION, METABOLISM, AND FERMENTATION
Polyols are often used as sugar substitutes in food products because they are difficult to digest and often slow to metabolize. This property is a result of the presence of the alcohol group in the area that is typically occupied by a carbonyl group in sugars [1]. It also prevents polyols from being hydrolyzed by salivary amylases and fermented by bacteria found in plaque, both of which contribute to the production of acid and caries. Smaller polyols such as monosaccharides are absorbed in the small intestine via passive diffusion along a concentration gradient (moving from high areas of concentration to low areas), whereas larger polyols (and polysaccharides) are too large to be absorbed in the gastrointestinal tract and are therefore poorly absorbed (less than 2% of oral intake is absorbed) [1].
If unabsorbed in the intestines, the polyols may be fermented by bacteria or excreted as is. The absorption and fermentation rates differ from polyol to polyol due to the different chemical structures (saccharide type and molecular weight) therefore, they will result in different glycemic responses and gastrointestinal side effects [1]. Thus, the amount of digestible carbohydrates that can also be metabolized in a food product is of interest to both food manufacturers and consumers.
NET CARBOHYDRATES
Currently, the USDA requires that food manufacturers label the total amount of carbohydrates in their products, including the total amount of sugar alcohols. However, some manufacturers believe that the total carbohydrate content on a food label can be misleading since not all carbohydrates in the food product are equally bioavailable.
This has led to some manufacturers devising a calculation known as “net carbohydrates,” which are meant to take into account how many digestible/ absorbable carbohydrates are in the product, informing consumers how many carbohydrates they are metabolizing [4]. Many manufacturers, aware that sugar alcohols tend to be poorly absorbed or metabolized, subtract the amount of sugar alcohols in the product from the total carbohydrate content, yielding the total “net carbohydrates.” Although this calculation is a large improvement over treating all carbohydrates as being equal, it too has been mindlessly calculated and has been incredibly misleading. For example, many food manufacturers will treat all sugar alcohols as being equal in their bioavailability, and thus, will either fully subtract them from the total carbohydrate content, or subtract 50% of their weight from the total carbohydrate content.
However, this is contradicted by the food science literature, which has shown for decades that these compounds exert different effects on the body [1]. For example, erythritol is fully absorbed within the small intestine, so it does not reach the colon and cause any gastrointestinal side effects, and it is also unable to be metabolized by the liver, meaning that it is excreted as is and has little effect on the body. To treat it as being equal to other sugar alcohols such as xylitol or maltitol is problematic given that the latter polyols are able to be metabolized by the body, leading to rises in blood sugar, and are also fermented by gut bacteria, leading to potential gut discomfort for some. Therefore, it is unreasonable to assume that both erythritol and xylitol can fully be subtracted from the total carbohydrate content to yield equal net carbohydrates.
Proper deduction of sugar alcohol content from total carbohydrates requires examining the properties and effects of individual sugar alcohols on a case-by-case basis by looking at the food science literature. In this report, we examine the properties of common sugar alcohols, their absorption and fermentation rates, and provide some guidelines on how they should be treated when calculating net carbohydrates with some practical examples.
3 | CALCULATING DIGESTIBLE CARBOHYDRATES
ERYTHRITOL
There are 0.2 kilocalories found in one gram of erythritol, making it 95% less energy dense than table sugar. Most of the erythritol (90% of it that is ingested in grams) is absorbed in the small intestine, however, almost none of it is metabolized in the liver and it is excreted in the urine as is [1]. This resistance to digestion and metabolization is due to the presence of an alcohol group in the region typically occupied by a carbonyl group in sugars. Therefore, erythritol has little impact on blood sugar levels compared to other sugar alcohols [5].
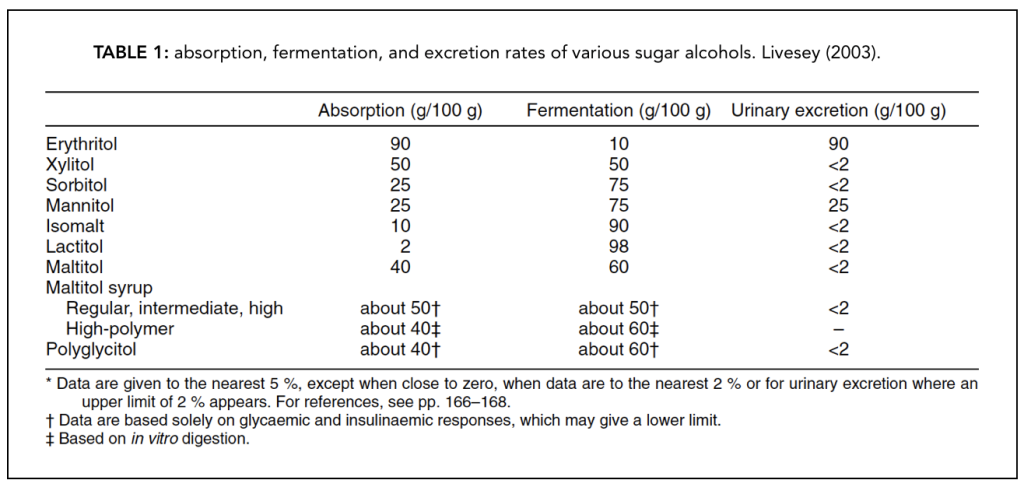
Further, less than 10% of it makes it to the large intestine thus, it is also the sugar alcohol that is the least likely to cause gastrointestinal side effects [6]. A table with these absorption, fermentation, and excretion rates can be found below [ Table 1]. The total grams of erythritol can be fully subtracted from the total carbohydrate since it is not metabolized, but this is one of the few exceptions.
MANNITOL
Mannitol has similar properties to erythritol in that any of it that is absorbed in the small intestine is eliminated from the body as is before it can be metabolized [1]. It differs slightly from erythritol, however, in that it can be fermented by the bacteria in the large intestine and is known to cause gastrointestinal side effects at dosages above 20 grams.
The total grams of mannitol can be fully subtracted from the total carbohydrate, just like erythritol.
SORBITOL
Only 25% of sorbitol is absorbed in the small intestine, while the rest is fermented by bacteria. The sorbitol that is absorbed in the small intestine can impact blood sugar levels because it is fully metabolized, although very slowly [1].
A good reasonable estimate would be to deduct 75% of the total grams of sorbitol from the total carbohydrates found in the food. For example, if there are a total of 20 grams of carbohydrates within the product, and 10 grams are from sorbitol, then we would deduct 7.5 grams from the total carbohydrate content, yielding about 12.5 or 13 grams of net carbohydrates.
XYLITOL
About 50% of the ingested xylitol in grams is absorbed in the small intestine while the rest is fermented by bacteria. It is also fully metabolized by the liver, but the end products (glucose or glycogen) are slowly released into the bloodstream [1]. A good reasonable estimate would be to deduct 65% of the total grams of xylitol from the total carbohydrates found in the food. For example, if there are a total of 20 grams of carbohydrates within the product, and 10 grams are from xylitol, then we would deduct 6.5 grams from the total carbohydrate content, yielding about 13.5 or 14 grams of net carbohydrates.
MALTITOL
When ingested, 40% of the maltitol gets absorbed in the small intestine and fully metabolized, while the rest is fermented (60%) is fermented by bacteria. However, unlike the other sugar alcohols (xylitol, sorbitol), maltitol is released into the bloodstream at a quicker rate than the others [1].
Therefore, a reasonable estimate would be to deduct 50% of the total grams of maltitol from the total carbohydrates that are found in food. With a total of 20 grams of carbohydrates within the product, and 10 grams coming from maltitol, we would subtract 5 grams from the total carbohydrate content, yielding 15 grams of net carbohydrates.
ISOMALT
Isomalt is very poorly absorbed in the small intestine (only 10 – 14 % is absorbed), while the rest is fermented by bacteria. However, this small absorbed amount can still impact blood sugar levels because it is metabolized fully and relatively quickly [1].
Therefore, a reasonable estimate would be to deduct 70% of the total grams of isomalt from the total carbohydrates that are found in food. If isomalt contributed 10 grams to the total carbohydrate content of a food product, then 7 grams of it would be subtracted to yield a total of 13 grams of net carbohydrates.
GLYCEROL / GLYCERIN
Glycerol or glycerin is fully metabolized by the liver and also fermentable by bacteria. It is rapidly absorbed, metabolized, and excreted by the body, therefore it can quickly impact blood serum levels of glucose and glycerol. However, unlike the other sugar alcohols, there are less available data that have explicitly looked at absorption, fermentation, and excretion rates [7].
A conservative estimate (based on the available data) would be to deduct 50% of the total grams of glycerol from the total carbohydrates that are found in food. So, if a product contained 20 grams of carbohydrates, and half came from glycerol, then like xylitol, 5 grams would be deducted from the total yielding a total of 15 grams of net carbohydrates.
While net carbohydrates are certainly an improvement over treating all carbohydrates as the same by using a sum score, many food manufacturers have also resorted to mindless calculations and deductions, often treating several food ingredients within a group as the exact same, defying much of the food science literature, which has consistently shown that different food products/ingredients contain unique matrices that affect the body differently. This can easily be seen by looking at the different biochemical structures of sugar alcohols which affect their absorption, fermentation, and excretion rates. Thus, it is critical to examine food ingredients and products on a case-by-case basis and determine what place they have within the diet.
4 | HEALTH OUTCOMES
As stated above, sugar alcohols provide several advantages as sugar substitutes due to the wide range of beneficial health effects they are associated with. Here, we examine some of these health effects, mostly within the context of randomized clinical trials, which are one of the most efficient ways to determine cause-and-effect relationships, assuming that the trials are free of any serious systematic errors in design or analysis.
ORAL HEALTH
The primary mechanism by which sugar alcohols minimize caries development is the lack of acidogenic potential. Thus, it is the absence of acid that results in caries prevention, which has been described by some researchers as a passive process rather than an active one [8]. Clinical trials have shown that xylitol, sorbitol, maltitol, erythritol, lactitol, and isomalt are noncariogenic [8–12].
However, sugar alcohols like xylitol have been found to be antibacterial against certain strains of bacteria such as S. mutans [11,13,14]. The reduction of such bacteria has been reported as contributing to a changed plaque ecosystem in which plaque quantity and adhesivity is reduced, which is significant given that plaque is a combination of bacteria and polysaccharides that contribute to the production of acid. Polyols have also been reported to reverse the formation of dental caries by accelerating remineralization and slowing demineralization of the tooth enamel by stimulating the flow of saliva [10,15,16].
In one systematic review and meta-analysis of five randomized controlled trials [17], xylitol administration in children was found to have a small reduction in dental caries, with higher doses of xylitol showing a larger effect. However, the quality of the evidence was low due to a small number of studies, the amount of between-study heterogeneity, and deficiencies in the designs of the primary studies. Furthermore, there was no quantification of the average effect across these studies.
In a systematic review and meta-analysis conducted by Cochrane [18], ten randomized controlled trials with a total of 5903 participants were analyzed and the results showed that fluoride toothpaste that contained 10% xylitol reduced dental caries by 13% in children and adults when compared to fluoride only toothpaste. In one of the studies included in the review, ingestion of high-dose xylitol syrup (8 grams) for a year showed a 58% reduction in dental caries in infants when compared to low-dose xylitol syrup. However, the analysis found that the overall quality of evidence was still very low and that more studies needed to be conducted.
A systematic review that compared non-fluoride interventions in reducing dental caries found that daily use of xylitol wipes in children may be an effective way of controlling dental caries, however, due to the limited data, the authors of the review were cautious with their recommendations [19].
RESPIRATORY HEALTH
It is estimated that there are nearly 17-18 billion cases of upper respiratory tract infections (URTIs) every year [20]. In more severe cases, they may lead to deaths; in 2014 there were approximately 3,000 mortalities caused by URTIs [21]. Such infections are typically caused by pathogenic microbes that colonize the upper airways [22]. Although the respiratory tract is occupied by commensal microbes that help prevent exogenous microbes from colonizing, many pathogenic organisms may still invade and colonize these sites during infections. Thus, new interventions that can help prevent and treat these infections are always sought out by researchers.
Xylitol interventions have shown much promise in reducing the bacterial counts of pathogens that typically cause these infections. In-vitro studies have shown that 5% xylitol solutions selectively reduce certain alpha hemolytic bacteria such as S. pneumoniae and that this effect is dose-dependent [23]. In one randomized crossover trial [24], 21 healthy volunteers were given 250 microliters of 5% xylitol and instructed to spray each nostril for four days. The main outcome that was assessed was nasal coagulase-negative Staphylococcus counts, which were reduced from 597 CFU/nasal swab during the control treatment with saline to 99 CFU/nasal swab during the xylitol treatment.
In another randomized controlled trial [25], 28 participants were randomized to either a xylitol nasal spray intervention or a control saline spray. The participants were instructed to use the sprays twice a day for 5 days. Although objective measures of rhinometry were not different between the groups, quality of life, as measured by the Rhinoconjunctivitis Quality of Life Questionnaire, indicated improvement for the xylitol spray group but not the control saline group.
The result from this study should be seen as being exploratory rather than confirmatory because the authors utilized statistical tests to see whether there was any improvement in the final scores for each group from baseline; however, this method of analysis is incorrect in randomized controlled trials because the main comparison should be a statistical test between groups that takes baseline scores into account as a covariate. Looking to see whether each group has any statistically significant improvements from baseline is especially problematic if there are imbalances between groups post-randomization, which could lead to regression to the mean and spurious effects [26,27].
Both animal models [28,29] and clinical trials have also suggested that xylitol may be useful for the treatment of sinusitis. In a clinical trial with 30 patients who suffered from chronic rhinosinusitis [30], nasal irrigation with a 5% xylitol solution for 30 days led to a reduction in Sino-Nasal Outcome Test 20 (SNOT-20) score when compared to the control group that used saline solution.
Acute otitis media (AOM) is a bacterial or viral infection of the middle ear that typically accompanies an upper respiratory tract infection [31]. It results in a painful and inflamed ear and can lead to temporary hearing loss. More severe infections that spread may even lead to impaired brain function. An important factor in the pathogenesis is the colonization of the upper airways by bacteria such as S. pneumoniae.
A Cochrane systematic review found five clinical trials that utilized xylitol interventions for the prevention and treatment of AOM in children up to 12 years of age [32]. The review concluded that there was moderate quality evidence suggesting that xylitol interventions may be effective in reducing the risk of AOM in children, but that it was not found to be effective for treating AOM during an upper respiratory tract infection. The authors also expressed caution in the interpretations of these data due to the limited number of studies that have been conducted.
DIABETES
Sugar alcohols have unique properties that make them a desirable sugar substitute for those who need to control their glycemic response. In one study, eight non-obese men fasted overnight and consumed 25 grams of lactitol, xylitol, or glucose [33]. The authors of the study used the indirect calorimetry method to measure carbohydrate and lipid oxidation in the participants after they consumed the solutions, and failed to find a significant difference in the participants that consumed lactitol or xylitol.
However, the results also suggest that the study may have been too small since glucose consumption resulted in a carbohydrate oxidation effect that was approaching statistical significance. Furthermore, another trial that recruited 10 obese and 10 lean, nondiabetic patients found that after eight hours of fasting, nasogastric administration of 50 grams of xylitol in water resulted in a very small increase in serum glucose when compared to placebo [34].
In a study where diabetic patients were provided single, 20-gram doses of erythritol, serum levels of erythritol reached their peak one hour after ingestion, however, there were no changes in serum glucose or insulin levels [5]. In a separate trial conducted by the same investigators but with different participants, daily administration of erythritol (20 grams) to 11 diabetic patients for 14 days resulted in a decrease in hemoglobin A1c and serum glucose levels. This is consistent with the properties of erythritol, which contribute to the inability of the body to metabolize it and lead it to being excreted by the body as is [1].
GASTROINTESTINAL HEALTH
It is well known that highly fermentable carbohydrates contribute to a healthy gastrointestinal ecosystem by increasing the diversity of the microbiome and by contributing to the production of various end products that maintain the integrity of the gastrointestinal tract.
Low molecular weight polyols, in particular, play an important role by reducing circulating levels of ammonia (NH3) [35] and other toxic microbial end products such as lipopolysaccharides, which are often associated with numerous metabolic and gastrointestinal disorders. Furthermore, polyols help maintain the acidic pH of the colon by contributing to the production of lactic acid and short-chain fatty acids such as butyric acid [36,37], which are a source of energy to colonocytes and help maintain the growth of acidophilic bacteria that typically reside in the colon.
In one animal study, the administration of xylitol to rats resulted in a shift from fecal gram-negative bacteria to gram-positive bacteria after six weeks when compared to animals not given xylitol [38]. Similar results have been found in crossover trials in humans, which reduce the sampling variability that results from individual and group differences. In one crossover trial, participants underwent an overnight fast and consumed a solution with 30 grams of xylitol or glucose in randomized orders [38]. Fecal microscopy analyses showed that glucose resulted in a 20-30% increase in gram-positive bacteria while xylitol resulted in a 50-55% increase.
The study also found that fecal levels of yeast were reduced, although, the species/strains were not reported [38]. However, in-vitro studies have suggested that sugar alcohols like xylitol are able to suppress the growth of yeast such as Candida [39], and other animal studies have shown xylitol to reduce the fecal concentration of Candida albicans [40]. Studies in rats have shown that xylitol consumption was associated with an increase in the genus Prevotella and the phyla Firmicutes while being associated with a reduction in Bacteroides and Clostridium [41,42].
Polyols have also been established as having utility for constipation due to their well-known effect of increasing osmotic pressure in the colon, thereby
increasing the retention of water and inducing a laxative effect, especially with higher dosages. In one randomized controlled trial [43], 120 participants were randomized to a xylitol chewing gum intervention or no chewing gum. When comparing the two groups, the xylitol chewing gum group had a reduced time in reaching the first bowel movement when compared to the control group (as measured by first bowel sounds). These results are consistent with other randomized trials that have been conducted on different populations such as women who had a caesarian section [44], which is often associated with constipation. Similar laxative effects have been reported for other sugar alcohols such as sorbitol, mannitol, isomalt, lactitol, maltitol, and erythritol.
ADVERSE EFFECTS
The most common adverse effects experienced by those who consume sugar alcohols are gastrointestinal effects. Side effects include bloating, diarrhea, and flatulence. However, the extent to which these side effects are experienced will differ from polyol to polyol and on the amount consumed [1,45].
Sugar alcohols that are poorly absorbed in the small intestine but highly fermentable in the colon will induce the most discomfort such as sorbitol, mannitol, and isomalt, whereas sugar alcohols that are more easily absorbed in the small intestine are less likely to induce such adverse effects with erythritol being the least likely to cause gastrointestinal adverse effects due to its high absorption rate (90%) [1,6].
Xylitol has been found in many studies to be better tolerated than polyol disaccharides, polysaccharides, and hexitols. This is consistent with the fact that xylitol is a monosaccharide that is more easily absorbed and less fermentable than larger polyol compounds [1]. However, a subset of individuals who chronically experience gastrointestinal discomfort, such as those with irritable bowel syndrome, a functional gastrointestinal disorder, have often been advised to avoid polyols and other complex forms of carbohydrates that are fermentable. One dietary approach that advocates this is the low-FODMAP diet.
While this diet has shown promise in many clinical trials [46], many of the trials are heterogenous and short in duration. Some critics of the diet have argued that although the diet may relieve symptoms temporarily, by consuming a diet devoid of fermentable carbohydrates, patients could potentially be worsening their health by reducing the diversity of their microbiome.
4 | DISCUSSION
It is quite clear that sugar alcohols possess several desirable properties that make them suitable as replacements for sugar. They are typically lower in calories, difficult to digest and metabolize quickly, and may even offer health benefits for several outcomes. As discussed above, xylitol has shown much promise for several health outcomes such as oral health, respiratory health, and metabolic conditions. It is even possible that other sugar alcohols may yield similar benefits given that they share several biochemical characteristics with xylitol. Although, more trials will need to investigate these compounds and their effects.
Regardless, there is no doubt that sugar alcohols are safe and effective sugar replacements for the vast majority of individuals.
5 | REFERENCES
[1] Livesey G. Health potential of polyols as sugar replacers, with emphasis on low glycaemic properties. Nutr Res Rev. 2003;16: 163–191. doi:10.1079/NRR200371
[2] Food and Agriculture Organization. Carbohydrates in Human Nutrition. Rome; 1998. Report No.: 66.
[3] Schiweck H, Bär A, Vogel R, Schwarz E, Kunz M, Dusautois C, et al. Sugar Alcohols. Ullmann’s Encyclopedia of Industrial Chemistry. American Cancer Society; 2012. doi:10.1002/14356007.a25_413.pub3
[4] Freeman J. The glycemic index debate: does the type of carbohydrate really matter? Diabetes Forecast. 2005;58: 11.
[5] Ishikawa M, Miyashita M, Kawashima Y, Nakamura T, Saitou N, Modderman J. Effects of oral administration of erythritol on patients with diabetes. Regul Toxicol Pharmacol RTP. 1996;24: S303-308. doi:10.1006/rtph.1996.0112
[6] Arrigoni E, Brouns F, Amadò R. Human gut microbiota does not ferment erythritol. Br J Nutr. 2005;94: 643–646. doi: 10.1079/bjn20051546
[7] Pelkonen R, Nikkilä EA, Kekki M. Metabolism of glycerol in diabetes mellitus. Diabetologia. 1967;3: 1–8. doi:10/d8qbs3
[8] Imfeld T. Efficacy of sweeteners and sugar substitutes in caries prevention. Caries Res. 1993;27 Suppl 1: 50–55. doi:10.1159/000261603
[9] Kawanabe J, Hirasawa M, Takeuchi T, Oda T, Ikeda T. Noncariogenicity of erythritol as a substrate. Caries Res. 1992;26: 358–362. doi:10.1159/000261468
[10] Featherstone JD. The science and practice of caries prevention. J Am Dent Assoc 1939. 2000;131: 887–899. doi:10.14219/jada.archive.2000.0307
[11] Mäkinen KK, Mäkinen PL, Pape HR, Peldyak J, Hujoel P, Isotupa KP, et al. Conclusion and review of the Michigan Xylitol Programme (1986-1995) for the prevention of dental caries. Int Dent J. 1996;46: 22–34.
[12] Rugg-Gunn AJ (University of N upon TDS. Lycasin and the prevention of dental caries. 1989 [cited 6 Jan 2020]. Available: http://agris.fao.org/agris-search/search.do?recordID=US9046966
[13] Wåler SM, Assev S, Rölla G. Xylitol 5-P formation by dental plaque after 12 weeks’ exposure to a xylitol/sorbitol containing chewing gum. Scand J Dent Res. 1992;100: 319–321. doi:10.1111/j.1600-0722.1992.tb01079.x
[14] Hayes C. The effect of non-cariogenic sweeteners on the prevention of dental caries: a review of the evidence. J Dent Educ. 2001;65: 1106–1109.
[15] Miake Y, Saeki Y, Takahashi M, Yanagisawa T. Remineralization effects of xylitol on demineralized enamel. J Electron Microsc (Tokyo). 2003;52: 471–476. doi:10.1093/jmicro/52.5.471.
[16] Leach S. Sugar substitutes and remineralization. Dtsch Zahnarztl Z. 1987;42: S135–S138.
[17] Marghalani AA, Guinto E, Phan M, Dhar V, Tinanoff N. Effectiveness of Xylitol in Reducing Dental Caries in Children. Pediatr Dent. 2017;39: 103–110.
[18] Riley P, Moore D, Ahmed F, Sharif MO, Worthington HV. Xylitol-containing products for preventing dental caries in children and adults. Cochrane Database Syst Rev. 2015; CD010743. doi:10.1002/14651858.CD010743.pub2
[19] Wang Y, Li J, Sun W, Li H, Cannon RD, Mei L. Effect of non-fluoride agents on the prevention of dental caries in primary dentition: A systematic review. PLoS ONE. 2017;12. doi:10.1371/journal.pone.0182221
[20] Vos T, Allen C, Arora M, Barber RM, Bhutta ZA, Brown A, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. The Lancet. 2016;388: 1545–1602. doi:10.1016/S0140-6736(16)31678-6
[21] Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. The Lancet. 2012;380: 2095–2128. doi:10.1016/S0140-6736(12)61728-0
[22] Brook I. Microbiology of chronic rhinosinusitis. Eur J Clin Microbiol Infect Dis Off Publ Eur Soc Clin Microbiol. 2016;35: 1059–1068. doi:10.1007/s10096-016-2640-x
[23] Kontiokari T, Uhari M, Koskela M. Effect of xylitol on growth of nasopharyngeal bacteria in vitro. Antimicrob Agents Chemother. 1995;39: 1820–1823. doi:10.1128/AAC.39.8.1820
[24] Zabner J, Seiler MP, Launspach JL, Karp PH, Kearney WR, Look DC, et al. The osmolyte xylitol reduces the salt concentration of airway surface liquid and may enhance bacterial killing. Proc Natl Acad Sci U S A. 2000;97: 11614–11619. doi:10.1073/pnas.97.21.11614
[25] Cingi C, Birdane L, Ural A, Oghan F, Bal C. Comparison of nasal hyperosmolar xylitol and xylometazoline solutions on quality of life in patients with inferior turbinate hypertrophy secondary to nonallergic rhinitis. Int Forum Allergy Rhinol. 2014;4: 475–479. doi:10.1002/alr.21311
[26] Bland JM, Altman DG. Best (but oft-forgotten) practices: testing for treatment effects in randomized trials by separate analyses of changes from baseline in each group is a misleading approach. Am J Clin Nutr. 2015;102: 991–994. doi:10.3945/ajcn.115.119768
[27] Vickers AJ, Altman DG. Statistics notes: Analysing controlled trials with baseline and follow up measurements. BMJ. 2001;323: 1123–1124. doi:10.1136/bmj.323.7321.1123
[28] Brown CL, Graham SM, Cable BB, Ozer EA, Taft PJ, Zabner J. Xylitol enhances bacterial killing in the rabbit maxillary sinus. The Laryngoscope. 2004;114: 2021–2024. doi:10.1097/01.mlg.0000147939.90249.47
[29] ain R, Lee T, Hardcastle T, Biswas K, Radcliff F, Douglas R. The in vitro effect of xylitol on chronic rhinosinusitis biofilms. Rhinology. 2016;54: 323–328. doi:10.4193/Rhin15.380
[30] Lin L, Tang X, Wei J, Dai F, Sun G. Xylitol nasal irrigation in the treatment of chronic rhinosinusitis. Am J Otolaryngol. 2017;38: 383–389. doi:10.1016/j.amjoto.2017.03.006
[31] Lieberthal AS, Carroll AE, Chonmaitree T, Ganiats TG, Hoberman A, Jackson MA, et al. The diagnosis and management of acute otitis media. Pediatrics. 2013;131: e964-999. doi:10.1542/peds.2012-3488
[32] Azarpazhooh A, Lawrence HP, Shah PS. Xylitol for preventing acute otitis media in children up to 12 years of age. Cochrane Database Syst Rev. 2016; CD007095. doi:10.1002/14651858.CD007095.pub3
[33] Natah SS, Hussien KR, Tuominen JA, Koivisto VA. Metabolic response to lactitol and xylitol in healthy men. Am J Clin Nutr. 1997;65: 947–950. doi:10.1093/ajcn/65.4.947
[34] Wölnerhanssen BK, Cajacob L, Keller N, Doody A, Rehfeld JF, Drewe J, et al. Gut hormone secretion, gastric emptying, and glycemic responses to erythritol and xylitol in lean and obese subjects. Am J Physiol-Endocrinol Metab. 2016;310: E1053–E1061. doi:10.1152/ajpendo.00037.2016.
[35] Blanc P, Daures JP, Rouillon JM, Peray P, Pierrugues R, Larrey D, et al. Lactitol or lactulose in the treatment of chronic hepatic encephalopathy: results of a meta-analysis. Hepatol Baltim Md. 1992;15: 222–228. doi:10.1002/hep.1840150209
[36] Mortensen PB, Holtug K, Rasmussen HS. Short-chain fatty acid production from mono- and disaccharides in a fecal incubation system: implications for colonic fermentation of dietary fiber in humans. J Nutr. 1988;118: 321–325. doi:10.1093/jn/118.3.321
[37] Clausen MR, Jørgensen J, Mortensen PB. Comparison of diarrhea induced by ingestion of fructooligosaccharide Idolax and disaccharide lactulose: role of osmolarity versus fermentation of malabsorbed carbohydrate. Dig Dis Sci. 1998;43: 2696–2707. doi:10.1023/a:1026659512786
[38] Salminen S, Salminen E, Koivistoinen P, Bridges J, Marks V. Gut microflora interactions with xylitol in the mouse, rat and man. Food Chem Toxicol. 1985;23: 985–990. doi:10.1016/0278-6915(85)90248-0
[39] Talattof Z, Azad A, Zahed M, Shahradnia N. Antifungal Activity of Xylitol against Candida albicans: An in vitro Study. J Contemp Dent Pract. 2018;19: 125–129. doi:10.5005/jp-journals-10024-2225
[40] Vargas SL, Patrick CC, Ayers GD, Hughes WT. Modulating effect of dietary carbohydrate supplementation on Candida albicans colonization and invasion in a neutropenic mouse model. Infect Immun. 1993;61: 619–626. Available: https://iai.asm.org/content/61/2/619
[41] Uebanso T, Kano S, Yoshimoto A, Naito C, Shimohata T, Mawatari K, et al. Effects of Consuming Xylitol on Gut Microbiota and Lipid Metabolism in Mice. Nutrients. 2017;9. doi:10.3390/nu9070756
[42] Tamura M, Hoshi C, Hori S. Xylitol affects the intestinal microbiota and metabolism of daidzein in adult male mice. Int J Mol Sci. 2013;14: 23993–24007. doi:10.3390/ijms141223993
[43] Gong Y, Zhang Q, Qiao L, Lv D, Ruan J, Chen H, et al. Xylitol Gum Chewing to Achieve Early Postoperative Restoration of Bowel Motility After Laparoscopic Surgery. Surg Laparosc Endosc Percutan Tech. 2015;25: 303–306. doi:10.1097/SLE.0000000000000174
[44] Lee JT, Hsieh M-H, Cheng P-J, Lin J-R. The Role of Xylitol Gum Chewing in Restoring Postoperative Bowel Activity After Cesarean Section. Biol Res Nurs. 2016;18: 167–172. doi:10.1177/1099800415592966
[45] Livesey G. Tolerance of low-digestible carbohydrates: a general view. Br J Nutr. 2001;85 Suppl 1: S7-16. doi:10.1079/bjn2000257
[46] Marsh A, Eslick EM, Eslick GD. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur J Nutr. 2016;55: 897–906. doi:10.1007/s00394-015-0922-1
Rafi, Z., Pendergrass, K. (2020) Absorption, metabolism, excretion, and health outcomes of various sugar alcohols. Ketogenic Diet Research. The Paleo Foundation.
Pendergrass, K., Rafi, Z. (2020)
Absorption, metabolism, excretion, and health outcomes of various sugar alcohols. Ketogenic Diet Research. The Paleo Foundation.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Rickaroons: a Case Study on Third-Party Paleo Certification
Rickaroons: a Case Study on Third-Party Paleo Certification
 Is Grain-Free the New Gluten-Free for Autoimmune Disease?
Is Grain-Free the New Gluten-Free for Autoimmune Disease?