Amyotrophic Lateral Sclerosis and the Keto Diet

First used in the 1920s, the ketogenic diet is most known for its established efficacy in treating intractable epilepsy and is best known clinically for this role. The mechanisms of the ketogenic diet are complex and not fully understood. Though, it likely involves metabolic adaptation in cellular signaling pathways, decreased neuronal excitability, and neuroprotection. As such, it is likely that the ketogenic diet may prove to be an effective treatment for other neurological diseases, including Manic Depressive Bipolar Disorder, GLUT1 Deficiency, and Amyotrophic Lateral Sclerosis (ALS). Here we discuss potential, though unconventional applications of the ketogenic diet.
While the aforementioned neurological diseases have little in common pathophysiologically, they each feature abnormalities in cellular energy metabolism. And, altered cerebral energy metabolism points to altered mitochondrial function. Nevertheless, an exploration into the therapeutic effects of a ketogenic diet on other neurological disorders is embryonic, at best. Therefore, their efficacy in treating disorders other than epilepsy in humans is speculative.
Amyotrophic Lateral Sclerosis (ALS) Symptoms
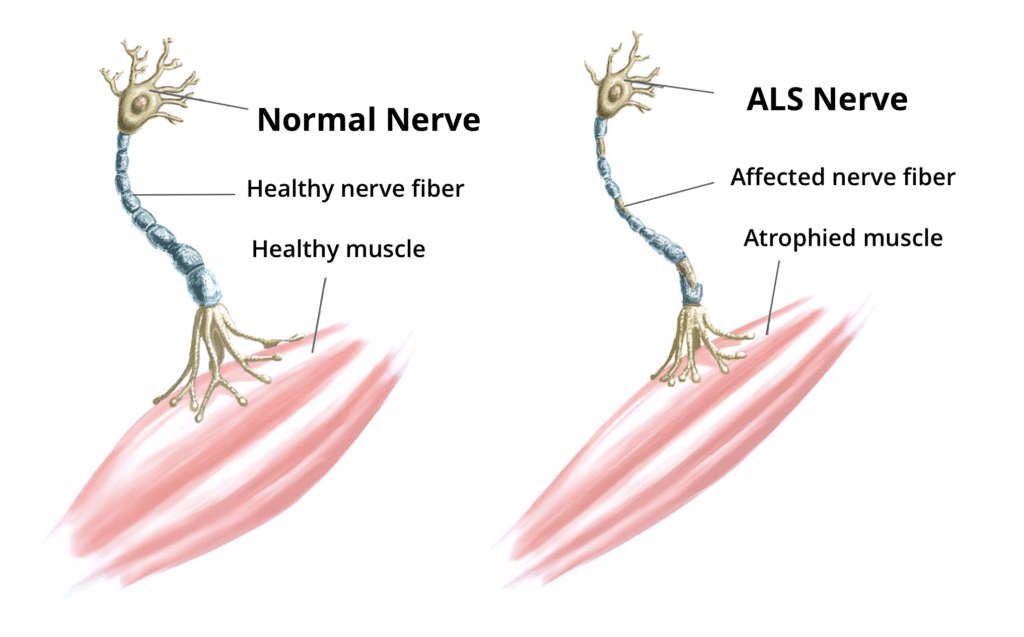
Amyotrophic lateral sclerosis (ALS) is a rapidly progressive disease due to degeneration of motor neurons of the cerebral cortex and anterior horn of the spinal cord that is universally fatal. It has claimed the lives of many, including physicist Stephen Hawking, heavyweight boxing champion Ezzard Mack Charles, and baseball legend Lou Gehrig who the disease is colloquially named after.
Most patients with ALS present with symptoms such as cramps, weakness, and muscle atrophy of the hands or feet. Later, weakness progresses to the forearms, shoulders, and lower limbs. Fasciculations, spasticity, hyperactive deep tendon reflexes, extensor plantar reflexes, clumsiness, stiffness, weight loss, fatigue, and difficulty controlling facial expression and tongue movements soon follow. Other symptoms include hoarseness, dysphagia, and slurred speech. Then, as swallowing becomes more difficult, patients often choke on liquids.
Ultimately, death is most often caused by the failure of the respiratory muscles. 50% of patients with ALS die within three years of the onset, 20% live for five years, and 10% will have a 10- year survival rate. Survival for longer than 30 years is extremely rare [1]. Stephen Hawking lived with ALS for 55 years.
In other words, ALS is a devastating degenerative disease with no known cure, and few treatment options exist. Treatments for ALS include glutamate blockers like riluzole, and muscle relaxers are given to reduce pain and discomfort. Like epilepsy, the exact pathological mechanisms that underlie ALS aren’t fully understood either. But what is known is that mechanistically, ALS likely involves oxidative damage, glutamate excitotoxicity, inflammation, and mitochondrial membrane dysfunction in nerve cells [2,3,4].
ALS and the Ketogenic Diet
Individuals with the disease have a mutation in the gene encoding copper/zinc superoxide dismutase (SOD1) [1]. So when a transgenic mouse model of ALS was created in which the gene SOD 1-G93A was overexpressed, it led to progressive muscle weakness and death resulting from respiratory failure, similarly to what occurs in humans with ALS. Studies on the mutant SOD 1-G93A mutant mice have given a lot of direction for future ALS studies, and treatment.
For example, in one study the SOD 1-G93A mutant mice were given a ketogenic diet and were found to have significantly higher motor neuron counts— and preserved motor function— compared to the control mice [5]. The authors also found that the mutant mice showed impaired electron transport chain function. However, administering the ketone body β-hydroxybutyrate restored activity in the neurons where electron transport chain function was either already decreased, or had been blocked pharmacologically [5]. While these results are indeed impressive, this study did not indicate an increase in lifespan of the mutant mice.
However, a more recent study found that the mutant mice fed a ketogenic diet variant called the Deanna Protocol delayed disease progression, extended the life expectancy in the ALS mouse model, and demonstrated improved motor function compared to controls [6]. These results are promising and carry the possibility that by raising ketones, the ketogenic diet and its variants may benefit patients with ALS.
References
1. Hulisz, D. (2018). Amyotrophic lateral sclerosis: disease state overview. Am J Manag Care, S320-S326.
2. Vucic, S., Rothstein, J. D., & Kiernan, M. C. (2014). Advances in treating amyotrophic lateral sclerosis: insights from pathophysiological studies. Trends in Neurosciences, 37(8), 433–442. doi:10.1016/j.tins.2014.05.006
3. Martin, L.J. (2011). Mitochondrial pathobiology in ALS. J Bioenerg Biomembr 43, 569–579.
4. Ari, C., Poff, A. M., Held, H. E., Landon, C. S., Goldhagen, C. R., Mavromates, N., & D’Agostino, D. P. (2014). Metabolic Therapy with Deanna Protocol Supplementation Delays Disease Progression and Extends Survival in Amyotrophic Lateral Sclerosis (ALS) Mouse Model. PLoS ONE, 9(7), e103526. doi:10.1371/journal.pone.0103526
5. Zhao, Z., Lange, D.J., Voustianiouk, A., MacGrogan, D., Ho, L., Suh, J., Humala, N., Thiyagarajan, M., Wang, J., and Pasinetti, G.M. (2006). A ketogenic diet as a potential novel therapeutic intervention in amyotrophic lateral sclerosis. BMC Neurosci 7, 29.
6. D’Alessandro G, Calcagno E, Tartari S, Rizzardini M, Invernizzi RW, et al. (2011) Glutamate and glutathione interplay in a motor neuronal model of amyotrophic lateral sclerosis reveals altered energy metabolism. Neurobiology of Disease. 43(2): 346–55.